
Licensable LNP Development Services
Proprietary Ionizable Lipid Platforms for High-Performing LNPs
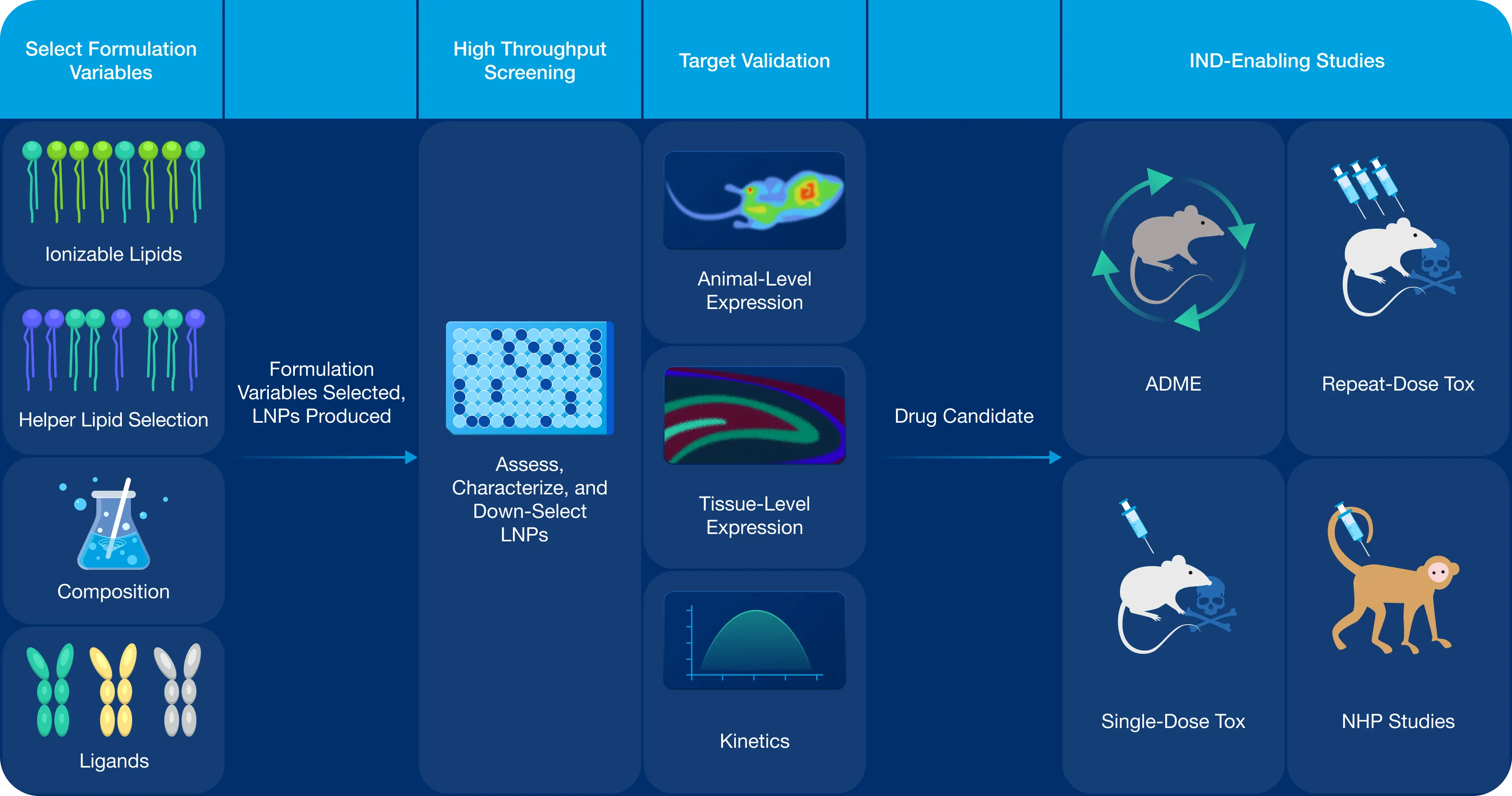
To complement the Phosphorex Drug Delivery Engine, we are honored to serve as NOF Corporation's North American LNP Center of Excellence, enabling evaluation of LNP candidates derived from proprietary, licensable ionizable lipid libraries. With the LNP Alliance, we support clients from lipid selection and LNP formulation through high-throughput screening, candidate nomination, and in vivo validation in rodent and non-human primate models.
In collaboration with the other members of the LNP Alliance, we offer integrated scientific expertise and a streamlined path through IND-enabling development.

Advanced Lipid Innovation Powered by NOF
NOF delivers advanced lipid technologies engineered for next-generation drug delivery. Its COATSOME® ecosystem features dual-responsive, ionizable lipids designed for efficient intracellular delivery with enhanced biosafety, combining endosomal membrane destabilization with reductive cytoplasmic release to enable synergistic payload transport.
Through its COATSOME® platform, NOF manufactures highly purified phospholipids and lipid derivatives for LNPs, liposomes, and emulsions, including PEGylated lipids for improved in vivo stability. They also offer custom lipid synthesis and novel lipid derivatives optimized for nucleic acid delivery, enabling tailored solutions for advanced therapeutic development.
Licensable LNP Development Solutions
For companies pursuing licensable LNP solutions, Phosphorex and NOF Corporation combine advanced lipid innovation with precision formulation expertise to design optimal, target-aligned LNPs. At our Hopkinton, MA, LNP Center of Excellence, we conduct high-throughput screening and iterative optimization to rapidly identify high-performing candidates.
Through the integrated capabilities of the LNP Alliance, we advance validated candidates into in vivo studies and IND-enabling development, delivering a streamlined, accelerated path to clinical readiness.

Ready to Engineer Particles for Precision Drug Delivery?
Partner with Phosphorex to transform your therapeutic vision into a precisely designed, clinically viable drug delivery solution.



